Welcome
sponsors/cros
Your Trusted Partner for Clinical Trials
Valencia Medical & Research Center offers experienced staff, robust infrastructure, and trusted community relationships to conduct high-quality Phase I-IV clinical trials across multiple therapeutic areas, ensuring reliable results.
Experienced Leadership
Our Principal Investigator, Dr. Inti Fernandez, and Sub-Investigator, Dr. Judith Valencia, are experienced MDs dedicated to participant safety, data integrity, and meticulous execution of trial protocols. Both investigators are passionate advocates for clinical research excellence. Their teamwork ensures consistently reliable trial outcomes.
Recruitment & Retention
We serve a diverse, predominantly Hispanic community with a large HIPAA-compliant database enabling quick, efficient participant recruitment. Our welcoming environment, dedicated team, and trusted MDs ensure strong relationships, high retention, and consistent trial data collection. Participant comfort remains our priority.
Research Quality Assured
At Valencia Medical & Research Center, we uphold the highest standards, consistently performing well in FDA audits without ever receiving a Form 483, and successfully passing sponsor evaluations. Our experienced team rigorously follows Good Clinical Practice (GCP), ensuring compliance, excellence, and readiness for future assessments.
COMMITMENT TO EXCELLENCE
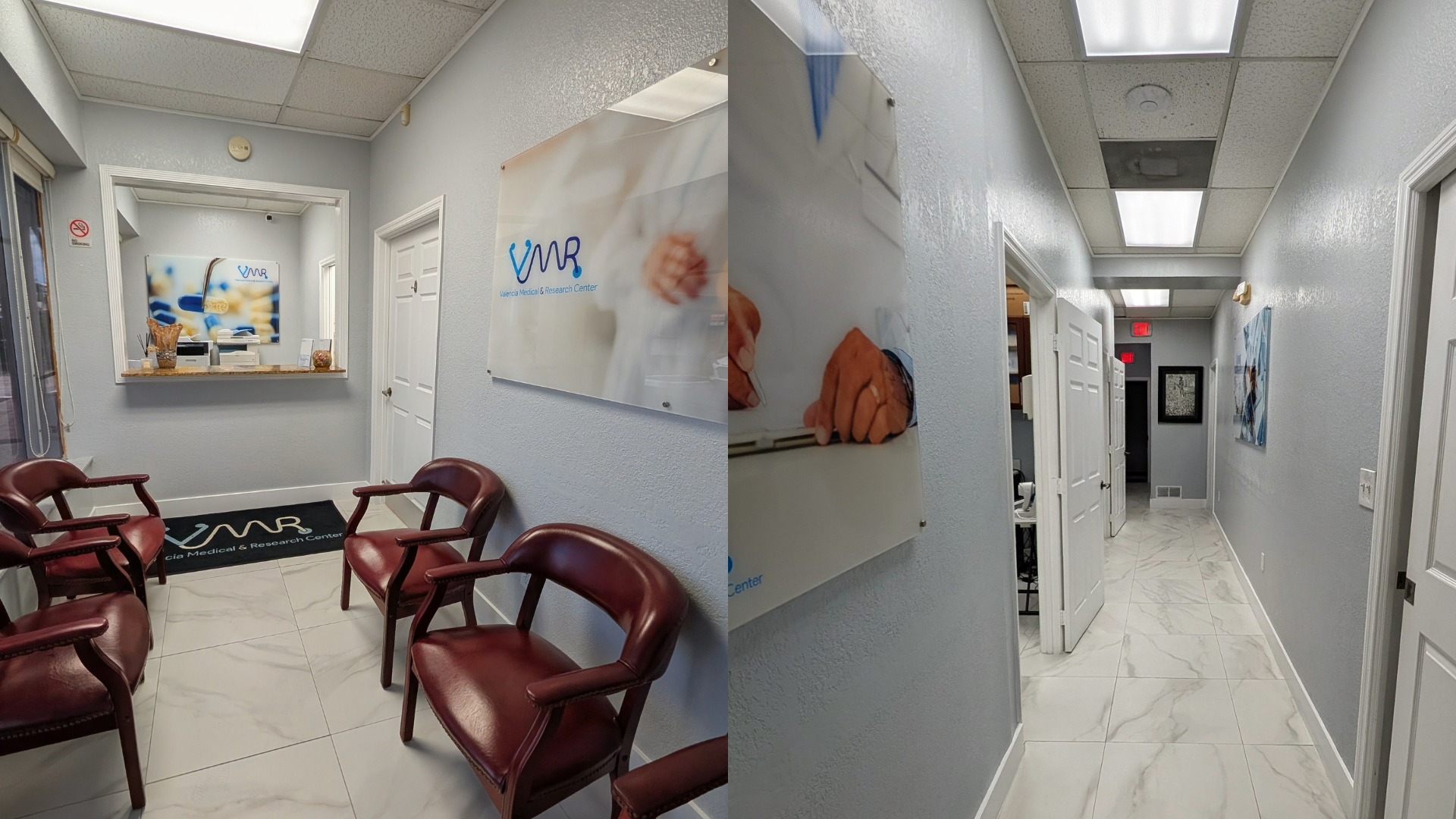
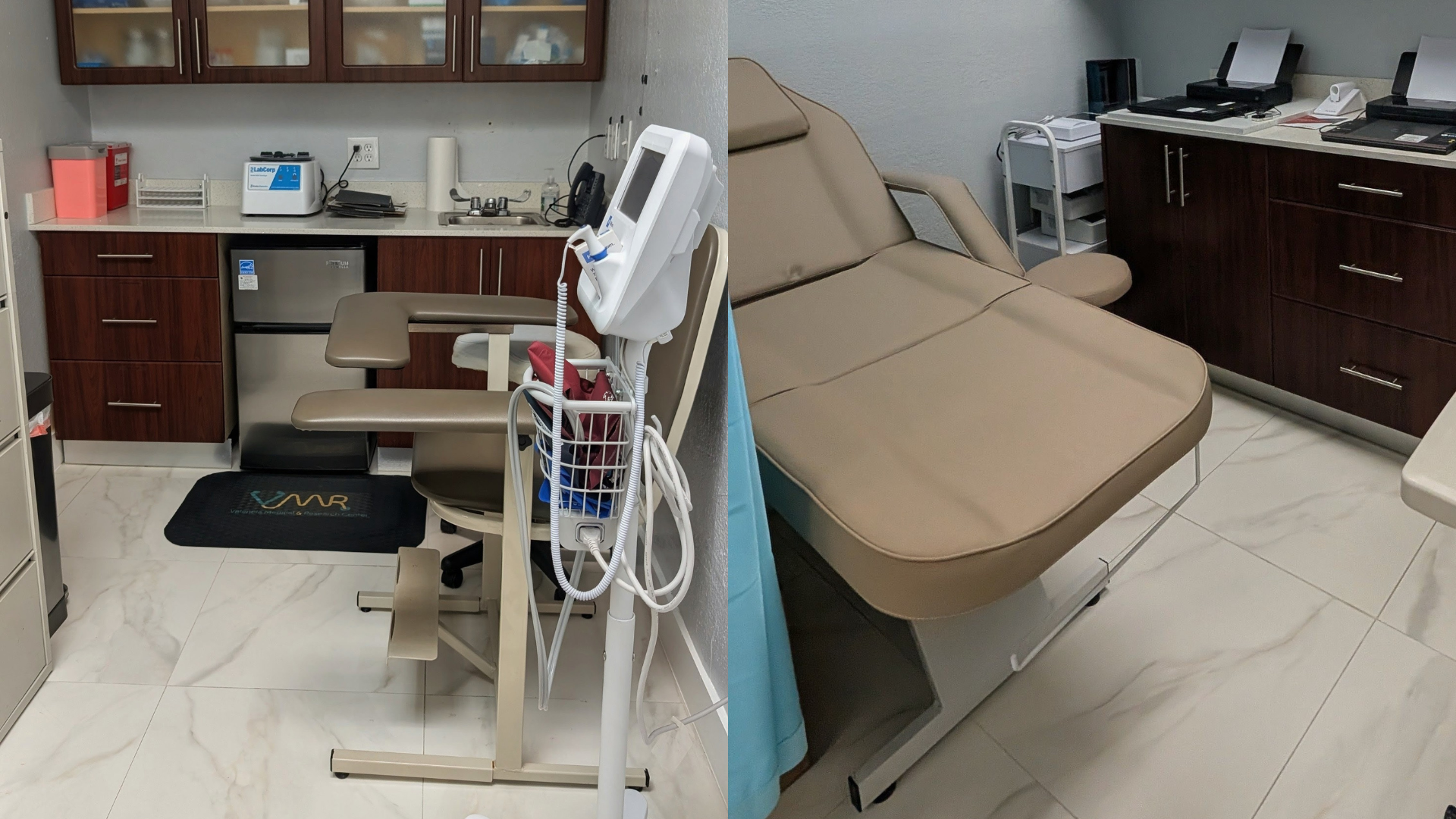
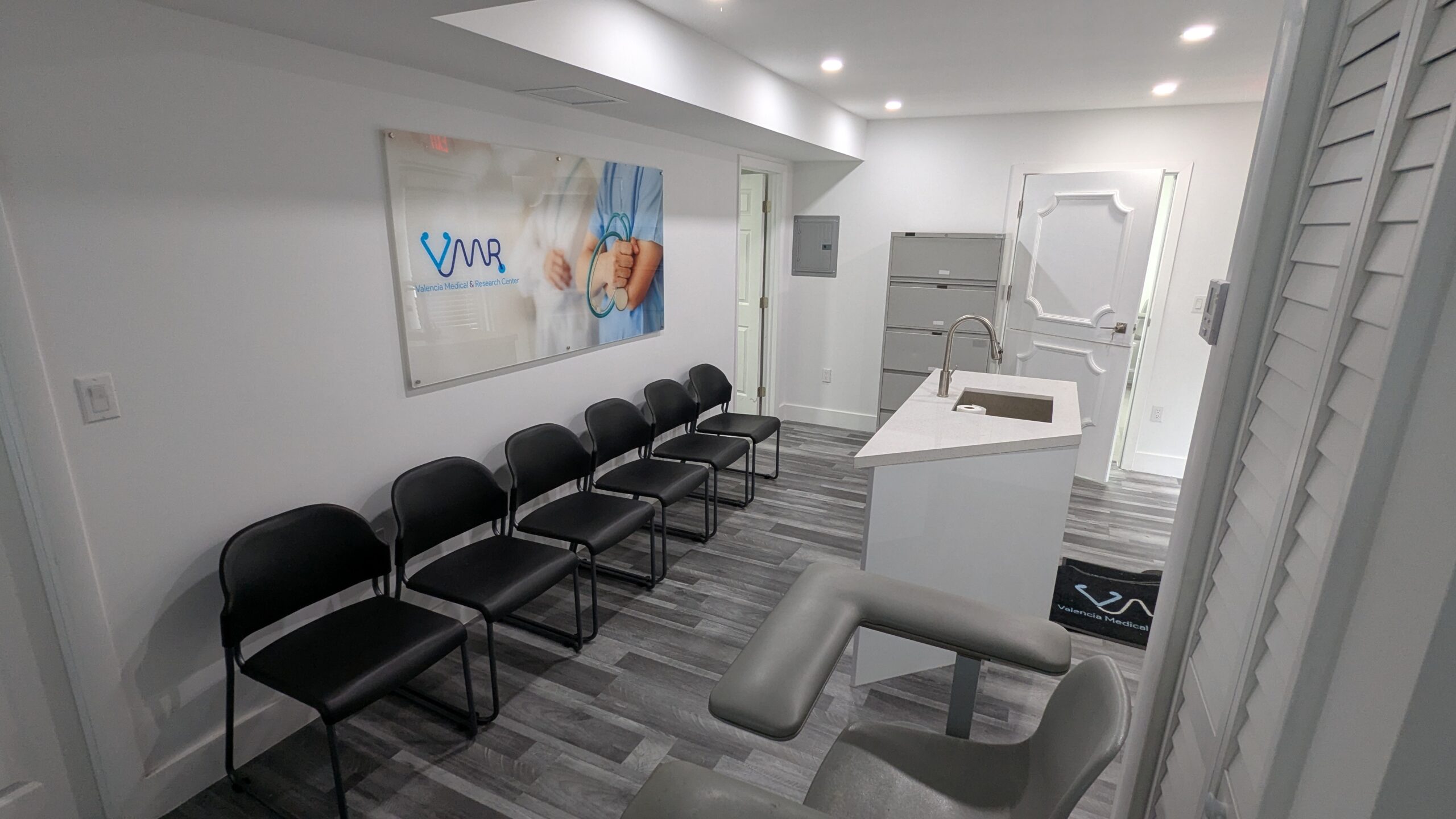
Our research center is integrated within a fully operational medical clinic, offering:
SPONSORS











CROS







Partner with us
Whether you’re a sponsor seeking a reliable research site or a CRO looking for an experienced partner, Valencia Medical & Research Center is equipped to support your clinical trials with a proven track record of compliance and success. We are committed to advancing medical research and ensuring the highest standards in patient safety and data accuracy.